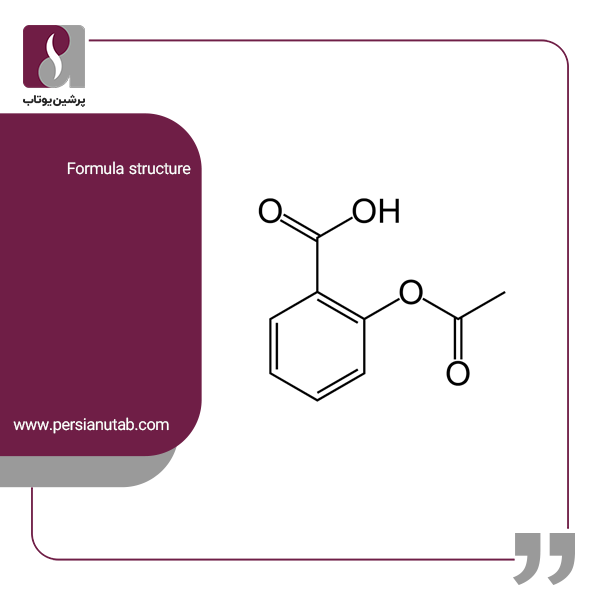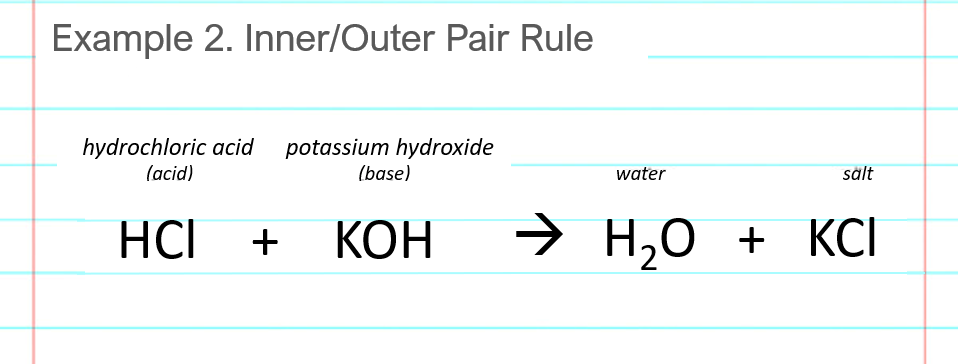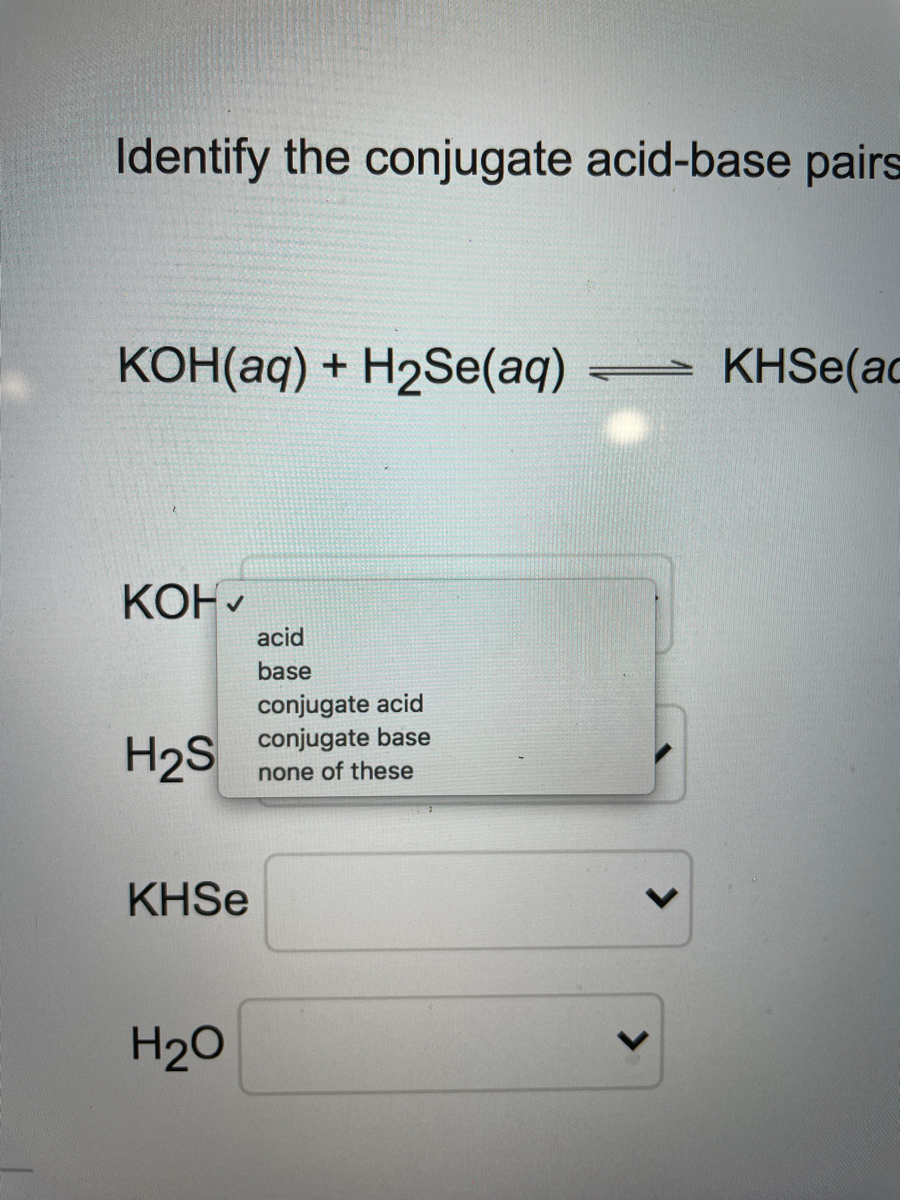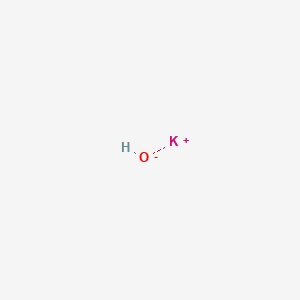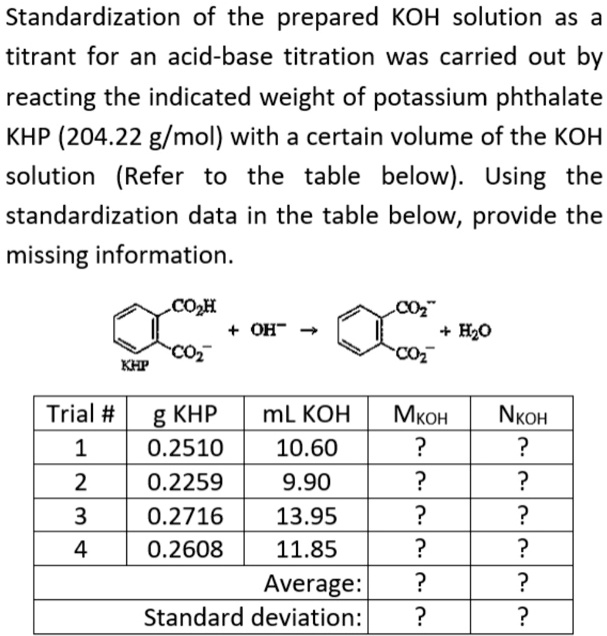
SOLVED: Standardization of the prepared KOH solution as titrant for an acid- base titration was carried out by reacting the indicated weight of potassium phthalate KHP (204.22 g/mol) with a certain volume of

Selective Focus of a Bottle of Pure Potassium Hydroxide or KOH Chemical Compound beside a Petri Dish with White Solid Pellets. Stock Photo - Image of flakes, base: 199192488

KOH/DMSO: A basic suspension for transition metal-free Tandem synthesis of 2,3-dihydroquinazolin-4(1H)-ones - ScienceDirect
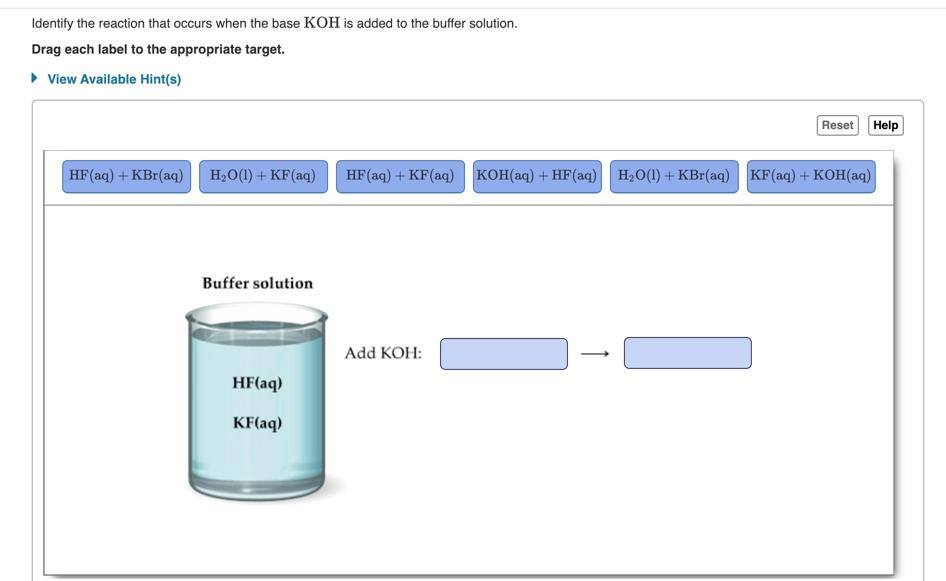
![Identify the reaction that occurs when the base KOH is added to the buffer solution. [{Image src='buffer_solution613505384566154416.jpg' alt='buffer solution' caption=''}] | Homework.Study.com Identify the reaction that occurs when the base KOH is added to the buffer solution. [{Image src='buffer_solution613505384566154416.jpg' alt='buffer solution' caption=''}] | Homework.Study.com](https://homework.study.com/cimages/multimages/16/buffer_solution613505384566154416.jpg)
Identify the reaction that occurs when the base KOH is added to the buffer solution. [{Image src='buffer_solution613505384566154416.jpg' alt='buffer solution' caption=''}] | Homework.Study.com
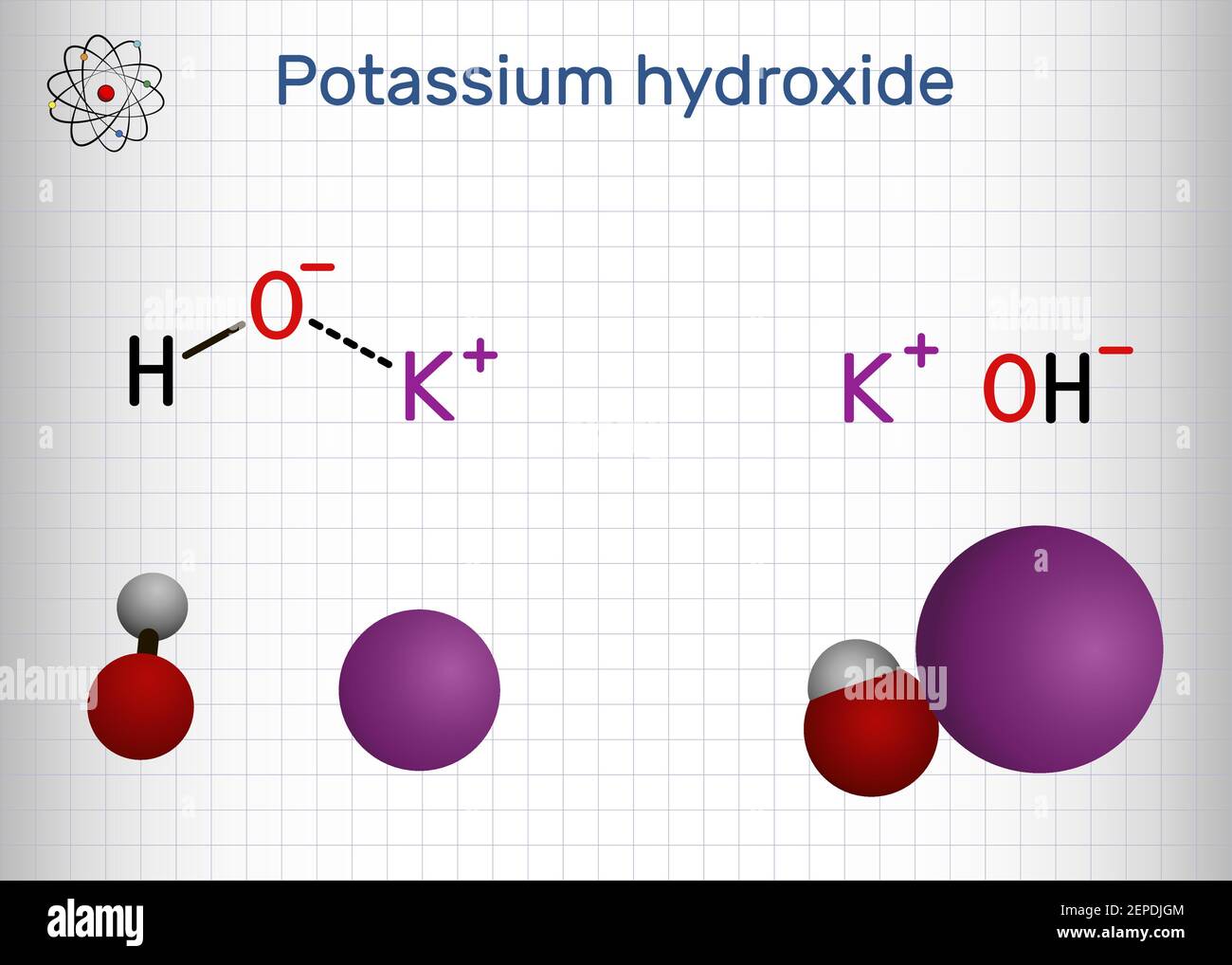
Potassium hydroxide, caustic potash, lye molecule. KOH is strong caustic base and alkali, ionic compound. Structural chemical formula and molecule mod Stock Vector Image & Art - Alamy


![Q. 2.41 Draw the products formed from th... [FREE SOLUTION] | StudySmarter Q. 2.41 Draw the products formed from th... [FREE SOLUTION] | StudySmarter](https://studysmarter-mediafiles.s3.amazonaws.com/media/textbook-exercise-images/image_vDzgYYT.png?X-Amz-Algorithm=AWS4-HMAC-SHA256&X-Amz-Credential=AKIA4OLDUDE42UZHAIET%2F20230701%2Feu-central-1%2Fs3%2Faws4_request&X-Amz-Date=20230701T162708Z&X-Amz-Expires=90000&X-Amz-SignedHeaders=host&X-Amz-Signature=d868f1850cb2c2f6ea1080c25b43bc82b032573ca0428417a162b1c05cd6e661)