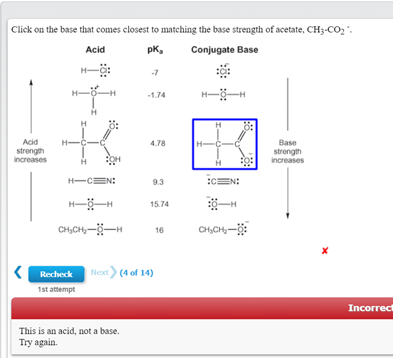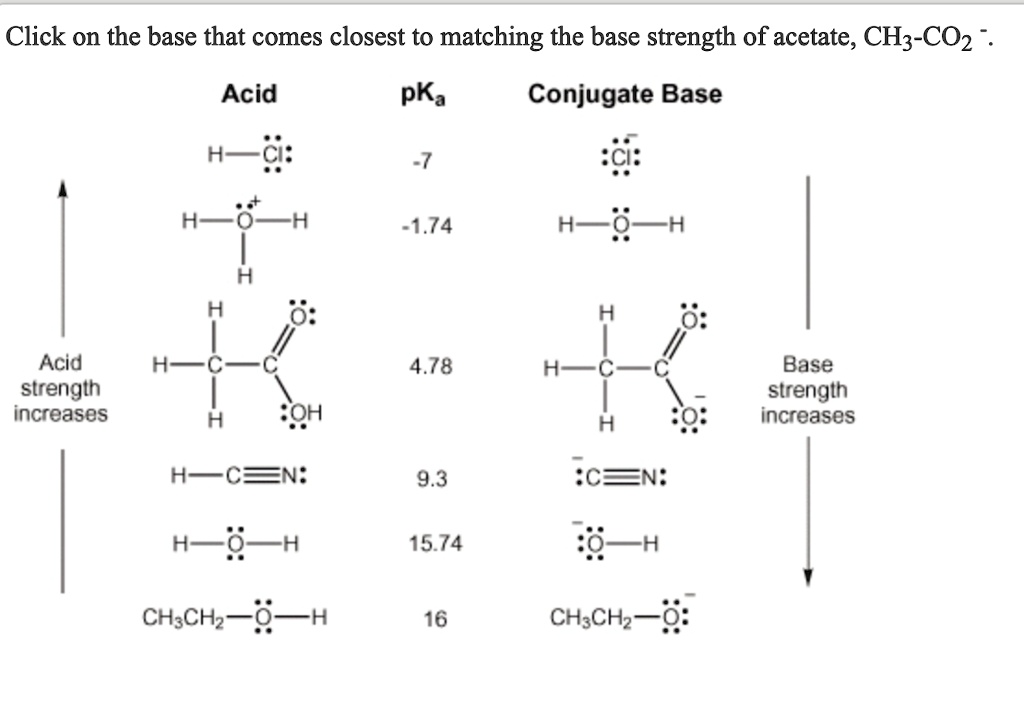
SOLVED: Click on the base that comes closest to matching the base strength of acetate, CH3CO2- Acid pKa Conjugate Base H3O+: 27 CIS H2O 21.74 H2O H H H Acid strength increases

SOLVED: For conjugate acid-base pairs, how are Ka and Kb related? Consider the reaction of acetic acid in water CH3CO2H(a q)+H2O(I) ⇌CH3CO2^-(a q)+H3O^+(a q) where Ka=1.8 × 10^-5 a. Which two bases
Why does the solution of sodium acetate give more concentration of Hydroxide ion? Shouldn't the number of Hydroxide ion and hydrogen ion be equal? - Quora
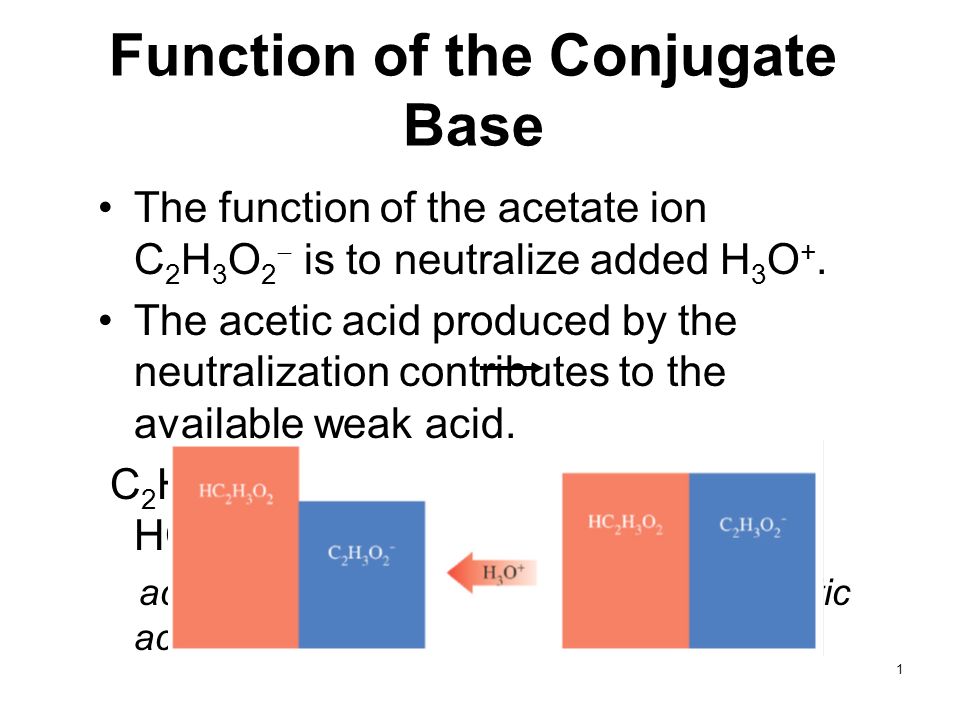
1 Function of the Conjugate Base The function of the acetate ion C 2 H 3 O 2 is to neutralize added H 3 O +. The acetic acid produced by the neutralization. - ppt download





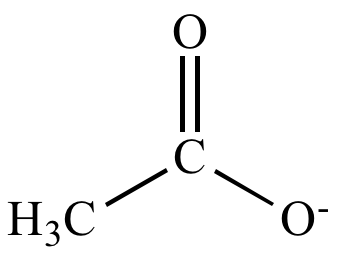



![Acetate [C2H3O2(−)] - Structure, Properties, Molecular Weight & Uses of Acetate ion. Acetate [C2H3O2(−)] - Structure, Properties, Molecular Weight & Uses of Acetate ion.](https://cdn1.byjus.com/wp-content/uploads/2019/04/structure-of-acetate.png)